Findings in clinical studies indicate that the combination of Raman Spectroscopy with multivariate data analysis, is the best fit for at-home non-invasive monitoring of glucose levels in people with diabetes. At RSP, we are not only demonstrating that Raman can measure glucose accurately and pain-free, but also that it provides stable calibration for a long period of time.
Key findings from our comprehensive clinical study, conducted at three clinical test sites [1], were published today in ACS Sensors under the headline: ‘Accurate post-calibration predictions for noninvasive glucose measurements in people using confocal Raman spectroscopy’’. The results presented in the article are based on a sample of 160 consecutive data subjects with manifested type 1 or type 2 diabetes, who used our device over the span of 41 days.
“These clinical results constitute a major breakthrough for non-invasive glucose monitoring in diabetes management. Stable calibration and good measurement accuracy are basic requirements for a non-invasive device to be used instead of a conventional, invasive CGM – and at RSP Systems, we are proud to share that we are finally there“, says Anders Weber, CEO of RSP Systems.
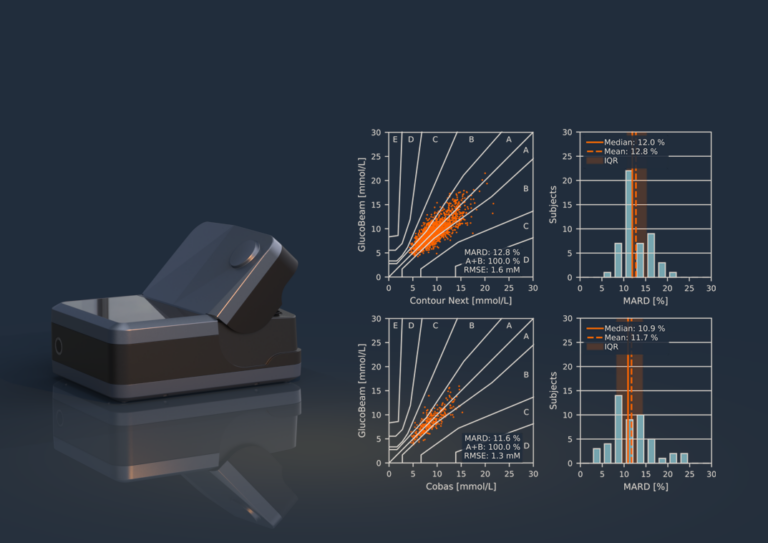
The article is written in collaboration with acknowledged leaders in the diabetes industry (Prof. Dr. Dietrich Hepp, Dr. Jan E. Henriksen and Dr. Guido Freckmann, MD) and reports the performance of GlucoBeamTM – a robust Raman-based, portable device for intermittent glucose determination – proven to be successful in the hands of lay people, irrespectively of age, gender, and skin colour.
“I am very impressed about the progress that RSP has made during the past 5 years in the development of non-invasive technology for diabetes management”, says Guido Freckmann, Medical Director and General Manager of the Institut für Diabetes-Technologies Forschungs- und Entwicklungsgesellschaft mbH.
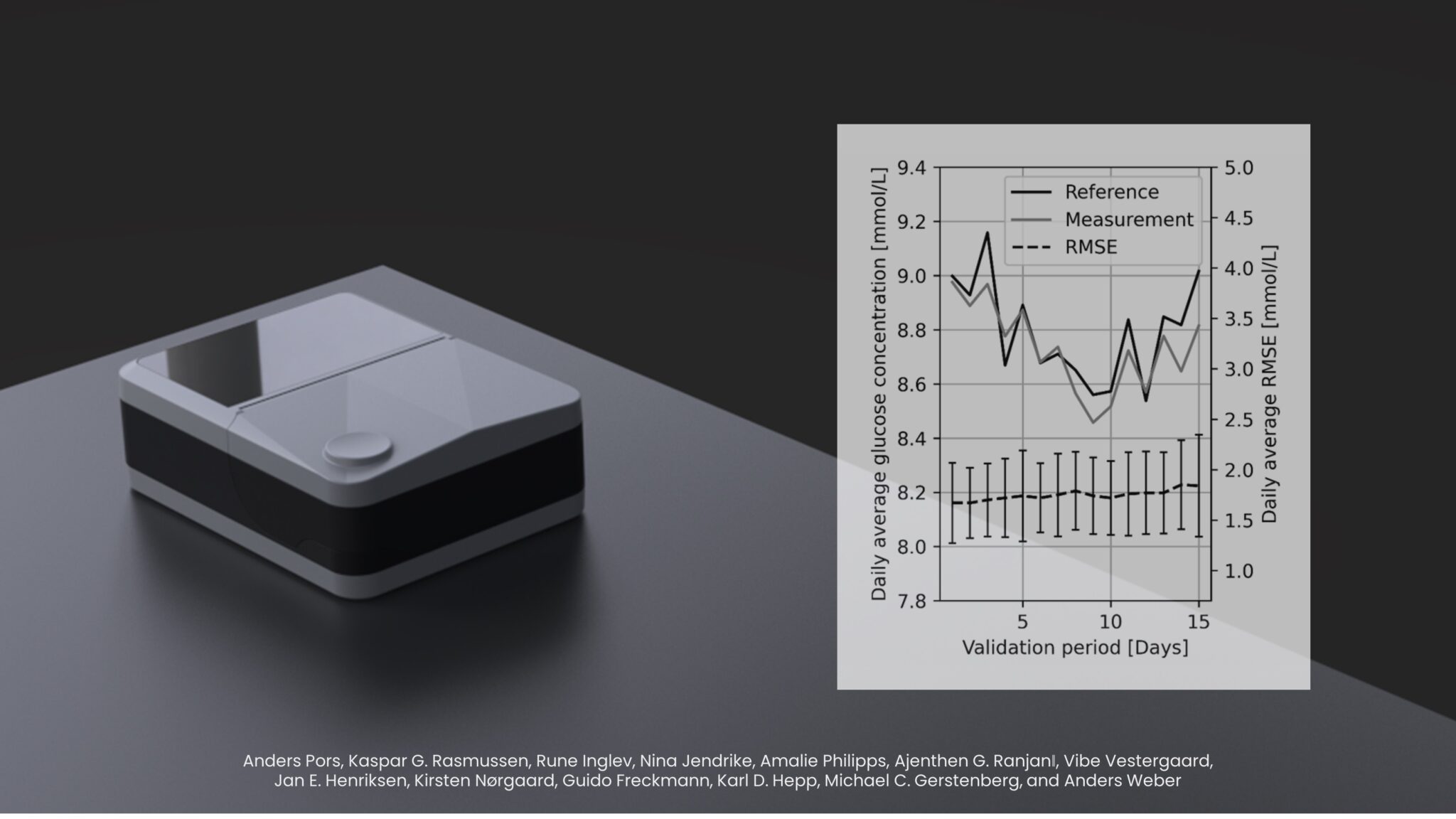
The study shows that the technology can be calibrated for practical, real-life usage, which in the article, is demonstrated through the measurement accuracy that remains stable over a validation period of 15 days – a major breakthrough that is the result of 15 years of research and development efforts by RSP Systems’ dedicated team of engineers and scientists. A subset of subjects with type 2 diabetes highlights very promising real-life results with a mean absolute relative difference (MARD) of 14.3 % in an uncontrolled home-setting, indicating accuracy on par with CGMs currently used for primary use in diabetes management.
These promising results, combined with our recently announced collaboration with Trumpf Photonics on the miniaturization of our technology are huge steps forward. We are excited to begin working on our new clinical study primo May 2023 in Ulm, Germany, which is currently far in the clinical trial authorization process. We look forward to keep on exploring the great possibilities that the future of non-invasive glucose monitoring technology holds.
You can access the publication on ACS Sensors website here.